 
A positively charged ion is called a cation. The green spheres represent negatively charged chloride ions and the gray. When an atom loses electrons, this results in a positive charge. When an atom gains electrons, this results in a negative charge. When an atom loses an electron to form a cation. Ions may be larger or smaller than the neutral atom, depending on the ion’s charge. 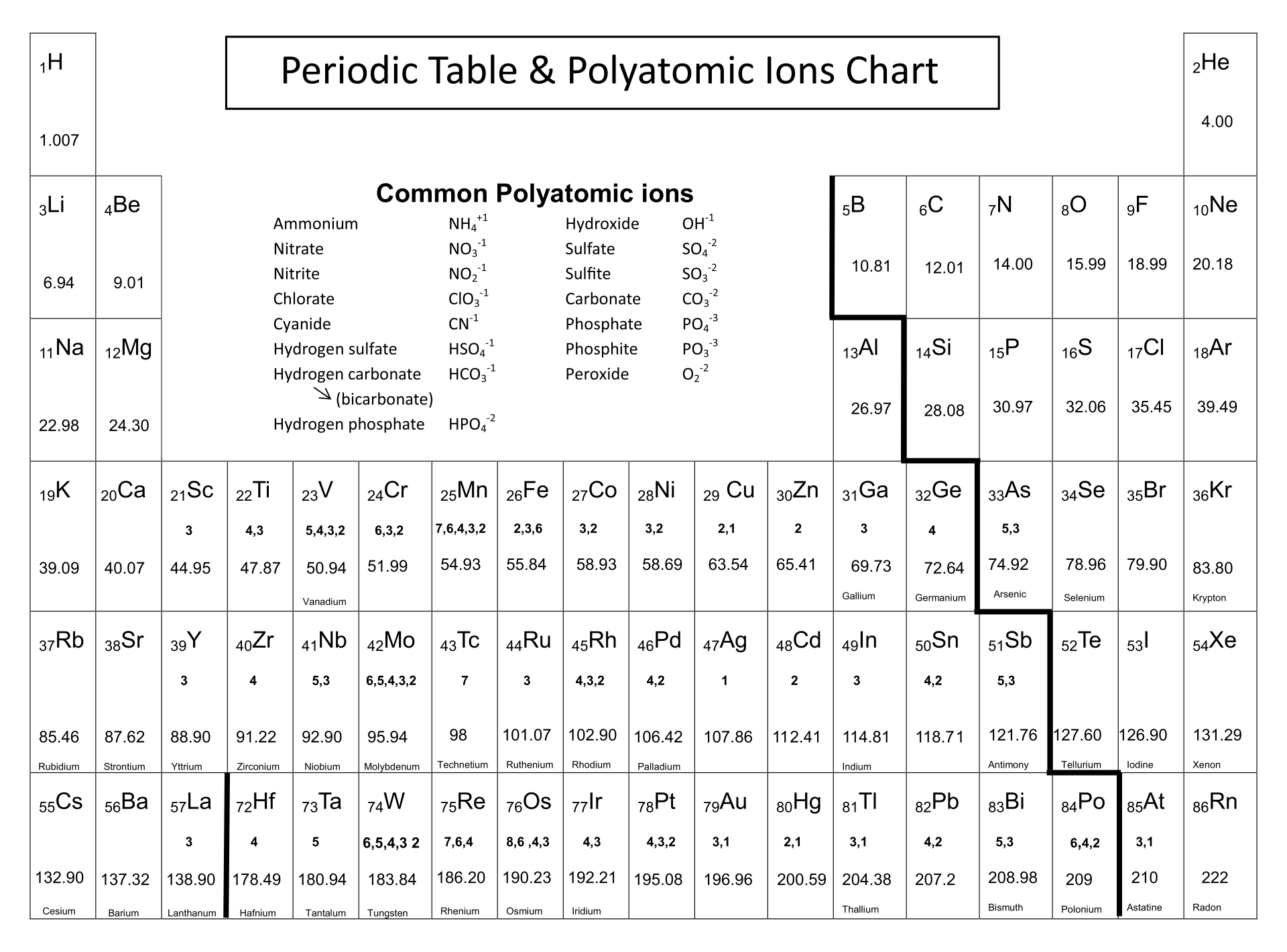
Ionic radius is the radius of an ion, regardless of whether it is an anion or a cation. For example, we would predict a carbon–chlorine distance of 77 pm + 99 pm = 176 pm for a C–Cl bond, which is very close to the average value observed in many organochlorine compounds. Ions, atoms with unequal amounts of electrons and protons, can have multiple types. Topic: The Periodic Table Variations Of Chemical Properties With Group And Row. If these values do indeed reflect the actual sizes of the atoms, then we should be able to predict the lengths of covalent bonds formed between different elements by adding them. This electric charge generated on the ion is known as Ionic charge. Here is my way of remembering which charge is with anion and cation: 'A' (in Anion) becomes before 'C' (Cation) in the alphabet. As mentioned above, there are positive and negative ions. In a similar approach, we can use the lengths of carbon–carbon single bonds in organic compounds, which are remarkably uniform at 154 pm, to assign a value of 77 pm as the covalent atomic radius for carbon. Ion - unequal amount of protons and electrons in an atom. Atoms of many main-group metals lose enough electrons to leave them with the same number of electrons as an atom of the preceding noble gas. The van der Waals radius is half the distance between chlorine nuclei in two different but touching Cl 2 molecules. You can use the periodic table to predict whether an atom will form an anion or a cation, and you can often predict the charge of the resulting ion. The covalent radius of Cl 2 is half the distance between the two chlorine atoms in a single molecule of Cl 2. An elements placement on the periodic table indicates whether its chemical charge is negative or. In many cases, the elements position on the periodic table will help you determine the kind of ion formed (anion or cation) and the size of the ionic charge. (d) This is a depiction of covalent versus van der Waals radii of chlorine. A chemical charge can be found by using the periodic table. (c) The van der Waals atomic radius, r vdW, is half the distance between the nuclei of two like atoms, such as argon, that are closely packed but not bonded. 
(b) The metallic atomic radius, r met, is half the distance between the nuclei of two adjacent atoms in a pure solid metal, such as aluminum. The alkali metals (shown in yellow) always form +1 ions. (a) The covalent atomic radius, r cov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as Cl 2. Many of the elements on the periodic table will always form ions that have the same charge. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
